 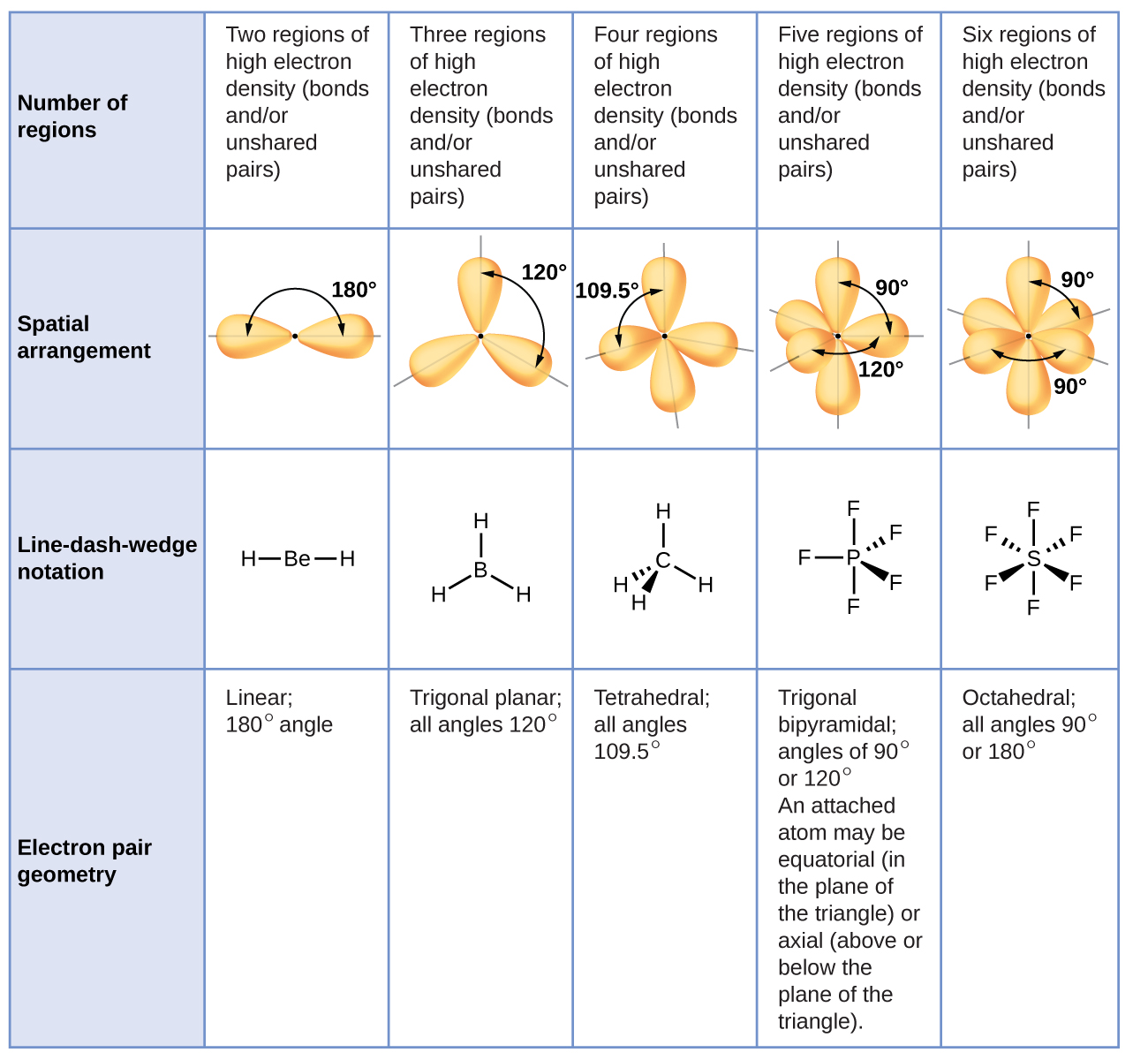
The ideal bond angles in a trigonal pyramid are based on the tetrahedral electron pair geometry. The primary difference is that with molecular geometry we include only the. The promotion of an electron from the 2 s orbital of beryllium to one of the 2 p orbitals is energetically uphill.\)) with the nitrogen atom at the apex and the three hydrogen atoms forming the base. An explanation of the difference between molecular geometry and electron geometry. The VSEPR theory predicts a molecule&039 s shape based on electron pairs around the central atom, considering the number of electron. The VSEPR chart confirms that the molecular geometry or shape of a molecule with an AX 2 generic formula is identical to its electron pair geometry i.e. The process of the formation of hybrid orbitals due to the intermixing of orbitals having similar energy levels is known as hybridization. 
The hybridization of boron in BF 3 is sp 2. In the BF 3 Lewis dot structure, a total of 9 lone pairs and 3 bond pairs are present. 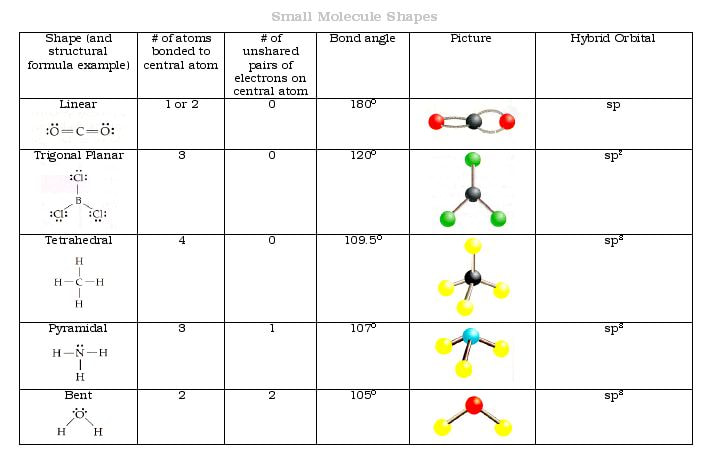
The AXN notation chart for different generic formulas is given below: CH3CN Hybridization. The electron geometry of BF 3 is also Trigonal planar, as its central atom, is surrounded by the 3 regions of electron density. \): A Hypothetical Stepwise Process for the Formation of BeH 2 from a Gaseous Be Atom and Two Gaseous H Atoms. the VSEPR shape of a molecule (hint: look at the VSEPR geometry chart). The electron geometry of the molecule is also linear with a bond angle of 180°. The electron-pair geometry provides a guide to the bond angles of between a terminal-central-terminal atom in a compound.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
